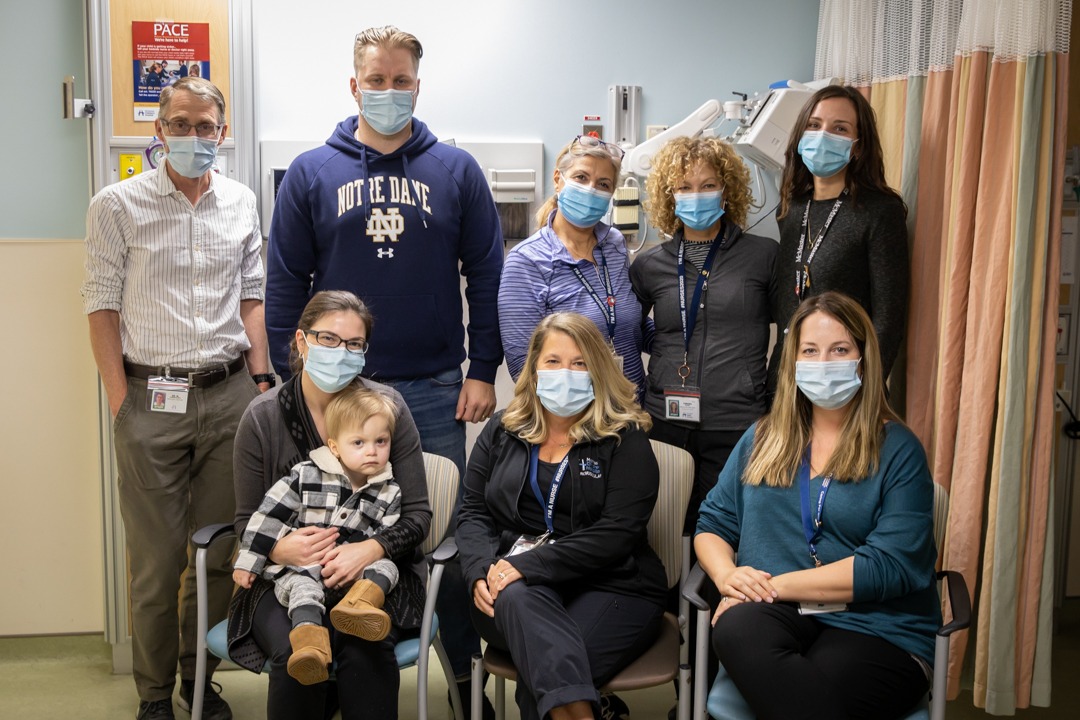
Family hopes March Break trip will be life-changing for son
After two years of staying close to home due to the pandemic, the O’Hagan family decided to travel over the March Break.
But instead of heading south for some sunshine, they drove east to Montreal so two-year-old Beau could take part in a clinical trial that his parents hope will improve his mobility.
Beau was born with spinal muscular atrophy (SMA), a genetic disease that damages nerves and leads to severe muscle weakness, impairing a person’s ability to move. Beau can push himself up and sit independently but he’s not able to walk.
Through the clinical trial, Beau received the drug Zolgensma — a form of gene therapy. This one-time treatment takes about one hour to administer by IV, and can work for up to 20 years. It has produced dramatic results, especially in babies under six months old. The trial is looking into whether older children may also benefit.
Spinal Muscular Atrophy
Nerves connect the spinal cord to muscles, signaling them to move. Spinal muscular atrophy happens when a gene called SMN-1 is missing or not working properly. Without this gene, the nerves begin to shut down, causing muscles to wither. This process starts in the embryo stage (before a baby is born) and continues in infancy, and is irreversible. It can affect a baby’s ability to hold their head up, sit up, roll over, crawl or walk. It can even impact breathing and swallowing, making the condition deadly in severe cases.

MCH patient Beau O’Hagan sits with his father Bryce at Montreal Children’s Hospital after undergoing gene therapy.
Type 1 SMA is the most severe. Without very early treatment, these children have a 50 per cent death rate by age two. Children with Type 2 are able to crawl, but can’t stand or walk independently. With the mildest form, Type 3, children are still weak but can move fairly well and can walk. Beau’s condition is Type 2. His parents hope that gene therapy will nudge him into the Type 3 category.
Newborn screening
Early diagnosis is the key to getting the best possible outcomes, says Dr. Mark Tarnopolsky, director of the Neuromuscular and Neurometabolic clinic at Hamilton Health Sciences’ McMaster Children’s Hospital (MCH).
While existing nerve damage can’t be undone, nerve and muscle deterioration can be stopped in its tracks with medication. The earlier children are treated, the better the results.
That’s why Ontario added SMA testing to routine newborn screening 18 months ago. Babies have a few drops of blood taken from their heel when they are about a day old. This blood is tested for more than 25 treatable diseases, including SMA.

Dr. Mark Tarnopolsky, director of the Neuromuscular and Neurometabolic Clinic at MCH.
Prior to SMA screening, babies and young children were diagnosed after failing to hit developmental milestones like rolling over, sitting up or standing. During the time lag between birth and diagnosis, irreversible damage continued to take place.
Treatment
There are currently three treatment options, with gene therapy being the newest on the scene.
“Dr. Tarnopolsky is the best person to have on your side.”
Gene therapy replaces a missing or faulty SMN-1 gene with a functioning gene. This treatment is now available in Ontario to children under six months old because studies show that’s where it has the most dramatic benefits. “We know that if gene therapy or one of the two other gene modifying treatments takes place in the first six months of a child’s life, it’s wildly beneficial,” says Tarnopolsky.
Beau took part in the clinical trial because he was too old to qualify for gene therapy in Ontario given that the studies have not yet had enough patients to determine whether or not it works in older children.

Beau O’Hagan, 2, can sit up independently but isn’t able to walk. Photo courtesy of Cecilia May Photography.
The other treatment options are medications called Spinraza and Risdaplam.
“It’s 100 per cent proven that the earlier children get any one of these three drugs, the better they’ll be…”
While the disease is caused by the loss of the SMN-1 gene function (leading to lower SMN1 protein), people also have a varying number of SMN-2 gene copies that can partially compensate for this loss by producing a somewhat less effective but still similar SMN-2 protein (the more copies a person has, the milder the disease). Both these medications increase SMN-2 gene expression.
Spinraza is delivered via a spinal injection every four months while Risdaplam – which Beau took before gene therapy – is a daily oral medication.
“It’s 100 per cent proven that the earlier children get any one of these three drugs, the better they’ll be because the nerves drop off so rapidly,” says Tarnopolsky, adding that his preference is gene therapy because the results are impressive – particularly in babies – and it’s less invasive because it’s a “one-and-done” treatment.
Beau’s future
As well as freeing Beau from taking daily medication, his parents hope that gene therapy will also improve his strength and mobility. “It’s our hope that this will help him someday walk,” says Rachel, who stayed in Montreal with Beau and his younger brother Rory for follow-up care and tests. Dad Bryce O’Hagan returned home to Beamsville to run the family business.
It’s possible to make physical gains because, even with permanent damage, the existing muscles can still grow and the central nervous system can become better coordinated – much like improvements gained by learning a sport. To help Beau progress, his parents work with him on a daily exercise regimen to help him build stregth and coordination. They even winterized their garage, adding an inflatable hot tub for year-round water therapy.
“Helping Beau has really sparked a passion in me for physiotherapy and occupational therapy,” says Rachel.
The family is grateful for the support they received from Dr. Tarnopolsky, Beau’s specialist, and the entire MCH team.
“Dr. Tarnopolsky is the best person to have on your side,” says Rachel, “because he’ll do whatever he feels is best and safest for your child.”
For more on Beau’s journey follow @BravelikeBeau on Instagram.